Last month, a colleague deferred a lip filler appointment after a 60-second AI-assisted dermoscopy scan flagged a suspicious lesion on the upper lip border. Biopsy later confirmed early melanoma.
That brief screen shifted the patient’s trajectory. It also reinforced what I treat as non-negotiable: no needles near skin until you’ve checked the treatment zone for lesion risk.
Cosmetic injectables can deliver predictable results when performed by trained, AHPRA-registered clinicians and supported by disciplined systems. The avoidable harms usually show up when screening, prescribing, documentation, or emergency readiness is treated as optional.
A safe injectable service depends on indication matching, lesion screening, compliant prescribing, and rehearsed complication response, every time.
Key Takeaways
Use these points to audit governance before you consent to treatment, or before you offer it in your clinic.
- Injectables work when governance works. Botulinum toxin reduces expression lines for roughly three to four months. Hyaluronic acid (HA) fillers restore volume for about six to eighteen months. Both require individualised consent, appropriate scope, and a written complication plan.
- Severe events are rare but high impact. Vascular occlusion is reported around 0.01 — 0.05% of filler cases. Filler-related blindness is estimated near 1 in 100,000. Hyaluronidase (for HA fillers) and an ocular emergency pathway should be immediately available.
- AI-assisted dermoscopy can strengthen pre-injection triage. Deep-learning models analysing dermoscopic images show sensitivity around 82% and specificity around 87% in pooled studies. Use AI for decision support, not diagnosis.
- Compliance is three-dimensional. The TGA restricts public advertising of Schedule 4 (S4) injectables. AHPRA requires a real-time prescriber consult for each S4 script. The OAIC requires explicit consent for clinical images and clear privacy handling.
- Choose providers who can prove the system. Verify registration, ask about lesion screening, and confirm emergency preparedness before you book.
Benefits: Where Injectables Deliver
Injectables deliver the best outcomes when you treat a specific indication with a clear timeline and a conservative plan.
Botulinum toxin targets dynamic rhytids, wrinkles that form with expression. Onset is usually three to seven days, peak effect is around one to two weeks, and duration is roughly three to four months (per Australian prescribing information).
HA fillers restore volume and contour in static folds, cheeks, and lips. Duration ranges from about six to eighteen months depending on product choice, depth, local motion, and individual metabolism (per Cosmetic Physicians College of Australasia guidance).
Document the expected onset, peak, and fade at consult. When patients understand timing and maintenance, they’re less likely to chase unsafe “top-ups” elsewhere.
Risks You Must Engineer Against
Most side effects are manageable, but the small number of severe complications demand systems, not reassurance.
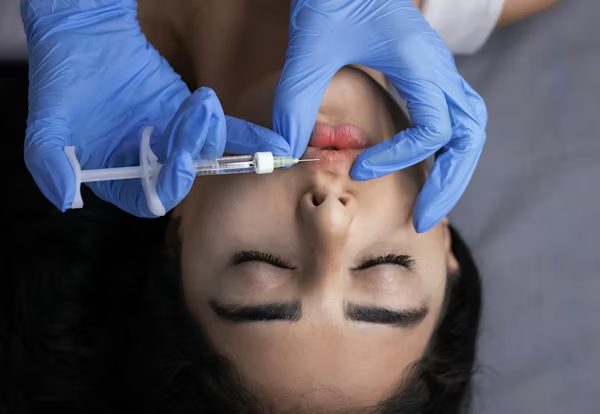
Expected short-term effects include injection pain, swelling, bruising, and temporary asymmetry. These usually resolve with time and aftercare.
Vascular occlusion after dermal filler is estimated at 0.01–0.05% across systematic review data, yet it can cause skin necrosis and permanent scarring. RANZCO (Royal Australian and New Zealand College of Ophthalmologists) guidance cites filler-related blindness near 1 in 100,000, with higher-risk areas including the nose, glabella, forehead, and nasolabial folds.
Prevention starts with anatomy competence and technique selection, including cannula use where appropriate. It also means slow, low-pressure injection, small aliquots, frequent aspiration where taught, and never injecting through or adjacent to a suspicious lesion.
On-site readiness should include in-date hyaluronidase, a high-dose “flooding” protocol (for suspected HA occlusion), warm compresses, clear escalation steps, and direct-dial contacts for ophthalmology and the ED (emergency department). If your team can’t run the first 10 minutes from memory, it isn’t ready.
Why AI-Assisted Dermoscopy Belongs in Aesthetic Clinics
Dermoscopy, a magnified and polarised skin examination, improves melanoma detection versus naked-eye inspection, and AI can strengthen triage when used correctly.
A 2024–2025 JAMA Dermatology meta-analysis across 100 studies supports dermoscopy’s added diagnostic accuracy. Across 27 studies of deep learning on dermoscopic images, pooled performance reached sensitivity around 82%, specificity around 87%, and AUC around 0.92 (area under the ROC curve).
In practice, many aesthetic patients are overdue for skin checks. A quick dermoscopy spot-check at consult can flag lesions at, or close to, planned injection sites and trigger referral before treatment.
Consumer phone apps that claim to “diagnose” skin cancer remain inconsistent in independent reviews. If you use AI, use clinician-grade tools with appropriate regulatory status, then document how AI informed, but did not replace, clinical judgment.
Building a Pre-Injection Screening Workflow
A lesion-safety gate is easiest to run when it’s a repeatable sequence with a clear stop rule.

- Intake: Ask the patient to point out any changing, bleeding, or non-healing spots, especially near planned injection sites. Record personal and family skin cancer history, immunosuppression, and anticoagulant use.
- Capture and review: Perform dermoscopy with appropriate lighting and focus, then add AI decision support if available. Confirm the software is regulated as SaMD (software as a medical device) where applicable.
- Apply a decision rule: Any “high-risk” AI flag, uncertainty, or clinician concern defers injections and triggers referral to a GP skin cancer clinic or dermatologist.
- Document: Store images, AI output, and your clinical decision in the EHR (electronic health record). Record what you told the patient and the agreed next step.
- Consent and privacy: Obtain explicit consent for photography and explain storage, retention, and who can access images. Avoid personal devices unless your governance covers metadata, access control, and secondary use.
In Australia, clinical images are usually treated as sensitive information under the Privacy Act. RACGP (Royal Australian College of General Practitioners) guidance also highlights practical risks with ad hoc photography, including unclear consent scope and uncontrolled data leakage.
How to Choose a Provider
A credible clinic can explain its safety system in plain language and show you how it’s implemented.
- Verify the injector’s AHPRA registration, scope, and any conditions.
- Confirm a prescriber consult happens in real time before each S4 prescription, not as a formality afterward.
- Ask what lesion screening is done near the planned injection site, and whether dermoscopy is used when indicated.
- Ask to confirm hyaluronidase is on site, in date, and paired with a written protocol and escalation pathway.
- Scan the clinic’s public messaging for compliance discipline, including no public promotion of prescription injectables.
If you’re in NSW and want a clinic that pairs skin-cancer know‑how with aesthetic services, it can help to review a provider’s scope, lesion-screening approach, prescribing pathway, photo-consent handling, and complication readiness in one place before you commit to an appointment or pay a deposit, especially if you’re new to injectables; the cosmetic injections page is a practical benchmark for what should be disclosed up front.
Compliance: The Australian Regulatory Trio
If you can’t meet AHPRA, TGA, and OAIC requirements at the same time, you don’t have a defensible injectable service.

AHPRA (Australian Health Practitioner Regulation Agency): The 2 September 2025 guidelines require a prescriber to conduct an in-person or video consultation each time S4 cosmetic injectables are prescribed. Asynchronous prescribing does not meet the standard. For patients under 18, botulinum toxin and fillers should not be prescribed for cosmetic purposes, and any exceptional pathway requires extra safeguards, including a minimum seven-day cooling-off period.
TGA (Therapeutic Goods Administration): Public advertising of prescription-only cosmetic injectables is prohibited. That includes language, pricing, or branding that effectively promotes an S4 medicine to the public. AI used for medical purposes, including dermoscopy decision support, is regulated as a medical device, so you need evidence of appropriate compliance and intended use.
OAIC (Office of the Australian Information Commissioner): Explicit consent is required for clinical photography. Your privacy notice should explain collection, storage location, retention period, access controls, and any secondary use, including marketing, education, or third-party platforms.
Frequently Asked Questions
These answers focus on practical decision points that reduce risk and improve outcomes.
Does AI-assisted dermoscopy replace a dermatologist?
No. Treat AI as decision support with a defined escalation threshold, and refer any high-risk flag or clinical concern for expert assessment.
Who can legally inject cosmetic injectables in Australia?
Only AHPRA-registered clinicians practising within scope can inject. S4 injectables also require a prescriber consultation each time, conducted in person or via live video.
Can I advertise injectables on my website or social media?
Public advertising of S4 medicines is prohibited, and indirect promotion can also breach the rules. Keep public content education-led, and move prescribing discussions into a compliant consultation pathway.
How long do results typically last?
Botulinum toxin typically lasts about three to four months. HA fillers commonly last six to eighteen months, depending on product, placement, and individual metabolism.
What emergency equipment should every clinic have?
At minimum: in-date hyaluronidase (where HA fillers are used), printed occlusion response steps, warm compress capability, incident documentation, photography consent forms, and a rehearsed escalation pathway with direct contacts for ophthalmology and the ED.



